Precipitation Reactions of Hydrated Ions
When examining the hydrolysis reactions of metal cations and oxoanions, possible interactions between the cations and anions in solution were ignored. However, some cation and anion pairs form insoluble salts.
A salt is:
|
Measuring the solubilities of different salts reveals a number of patterns which are often referred to as "solubility rules". Students are encouraged to memorize these rules which can be employed to determine if a given salt will dissolve in water or precipitate from solution.
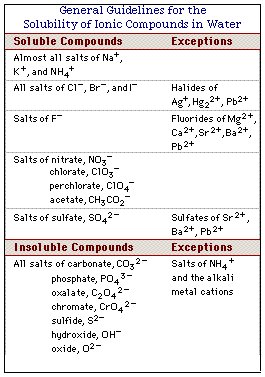
Text book listings of these rules do not cover all the possible cations and cover only a few anions.
Even chemical handbooks such as the CRC Handbook of Chemistry and Physics, which contains an extensive tabulation of the physical constants of inorganic compounds, cover only a fraction of the known inorganic salts.
Our goal will be to develop a general scheme for predicting the solubility of a wide variety of inorganic salts.
| yMm+ (aq) + mXy- |
Thermodynamically, whether precipitation occurs or not depends on the Gibbs Free Energy change, DGo.


|
To analyze the thermodynamics of precipitation reactions, both enthalpy and entropy changes must be considered.
- Weakly, moderately, strongly, very strongly acidic cations
- weakly, moderately, strongly,very strong basic anions


This generalized rule includes two of the traditional "solubility rules":
- all carbonates are insoluble except those of the Group 1A elements and NH4+
- all hydroxides are insoluble except those of the Group 1A elements, Sr2+, and Ba2+
- adds the salts of a number of additional oxo anions, such as phosphate, arsenate, silicate and borate
- predicts the behavior of cations not listed in the rules, i.e. TlOH,
Eu(OH)2 and Tl2CO3 are all soluble
Rule #2. Nonacidic cations and nonbasic anions give moderately insoluble salts.
- There aren't many nonacidic cations and nonbasic anions


Rule #3. Cross-combinations give soluble salts.

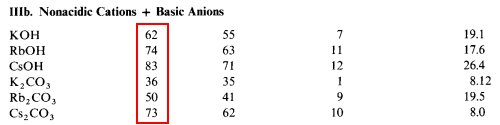

The rule that all nitrates are soluble falls into this category.
Rule #4.The solubility of feebly basic anions such as sulfate are difficult to predict.
- The tendency of these salts to precipitate or not precipitate varies with the relative magnitude of the opposing entropy and enthalpy terms
- Many feebly acidic cations and feebly basic anions give insoluble salts

The solubilities on insoluble salts of basic anions are enhanced in strongly
acidic solution.
|
Classify each of the following salts as soluble or insoluble in water and in strongly acidic solutions.
|