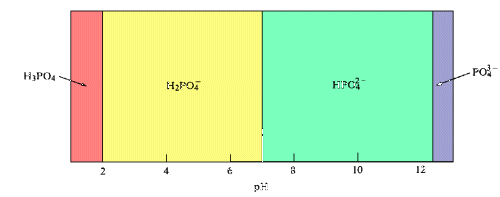
However, if suitable reducing agents are present, the phosphorous may not remain in the +5 oxidation state.
Knowledge of the pH condition of the environment is not sufficient for predicting the form in which an element will exist in natural waters. You must also take into consideration whether the aqueous environment is well aerated (oxidizing) or polluted with organic wastes (reducing). In order to add this variable, we must expand the predominance diagram to include the reduction potential of the environment as well as the pH. This type of predominance diagram is known as a Pourbaix diagram.Eo-pH diagram, or pE-pH diagram.
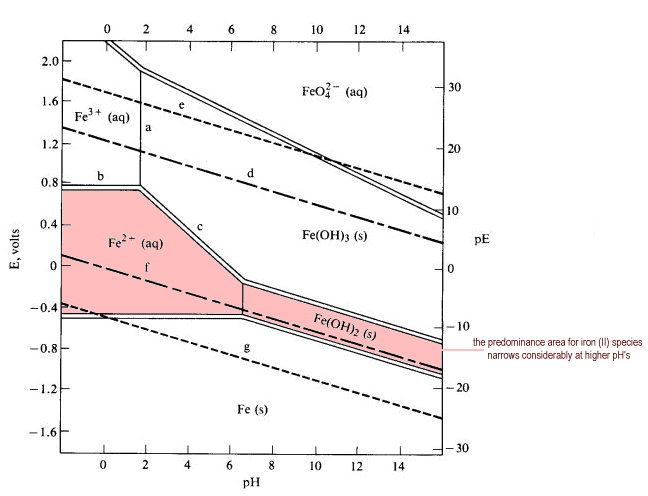
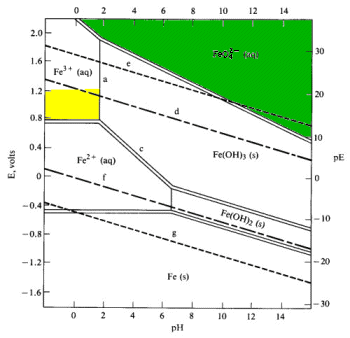
Simplified Pourbaix diagram for 1 M iron solutions.
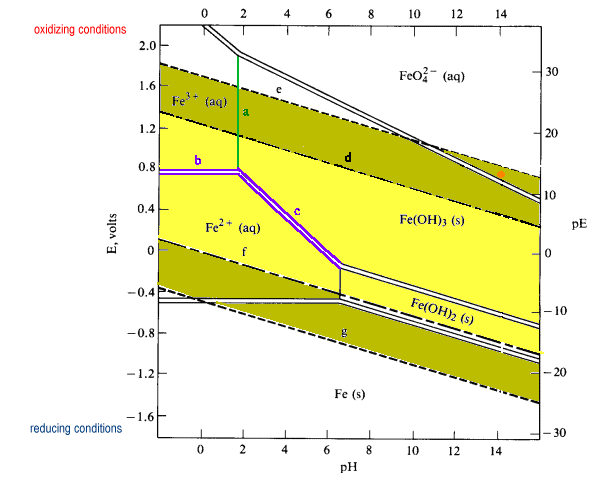
Low E (or pE) values represent a reducing environment. High E values represent an oxidizing environment. The pE scale is intended to represent the concentration of the standard reducing agent (the e-) analogously to the pH scale representing the concentration of standard acid (H+). PE values are obtained from reduction potentials by dividing Eoby 0.059.
Key to features on the diagram:
- Solid lines separate species related by acid-base equilibria (line a)
- line a shows the pH at which half of the 1 M iron is Fe3+ and half is precipitated as Fe(OH)2
- Pourbaix diagrams incorporate Z1/r calculations and acid-base equilibria
- the position of an acid-base equilibrium is dependent on the total concentration of iron
- reducing the total concentration of Fe3+ will reduce the driving force of the precipitation
- reducing the total iron concentration from 1 M to 10-6 M (more realistic concentrations for geochemists and corrosion engineers) shifts the boundary from pH 1.7 to pH 4.2
- In general, in more dilute solutions, the soluble species have larger predominance areas.
- Solid double lines separate species related by redox equilibria (lines c & d)
- redox equilibria of species not involving hydrogen or hydroxide ions appear as horizontal boundaries (line b)
- redox species of species involving hydrogen or hydroxide appear as diagonal boundaries becuase they are in part acid-base equilibria (line c)
- diagonal boundaries slope from upper left to lower right because basic solutions tend to favor the more oxidized species
- Longer dashed lines enclose the theoretical region of stability of the water to oxidation or reduction ((lines d & f) while shorter dashed lines enclose the practical region of stability of the water (e & g)
- Dashed line d represents the potential of water saturated with dissolved O2at 1 atm (very well aerated water).
- above this potential water is oxidized to oxygen:
2 H2O + 4 H+ (aq) O2 + 4 e- Eo = +1.229 V - theoretically water should be oxidized by any dissolved oxidizing agent Eo > 1.229
- in practice, about 0.5 V of additional potential is required to overcome the overvoltage of oxygen formation (dashed line e)
- Dashed line f represents the potential of water saturated with dissolved H2 at 1 atm pressure (high level or reducing agents in solution).
- Below this potential water is reduced to hydrogen:
2 H+ + 2 e- Eo = +1.229 V - in practice, an overvoltage effect prevents significant release of hydrogen until the lower dashed line g is reached
 Uses of Pourbaix Diagrams:
Uses of Pourbaix Diagrams:
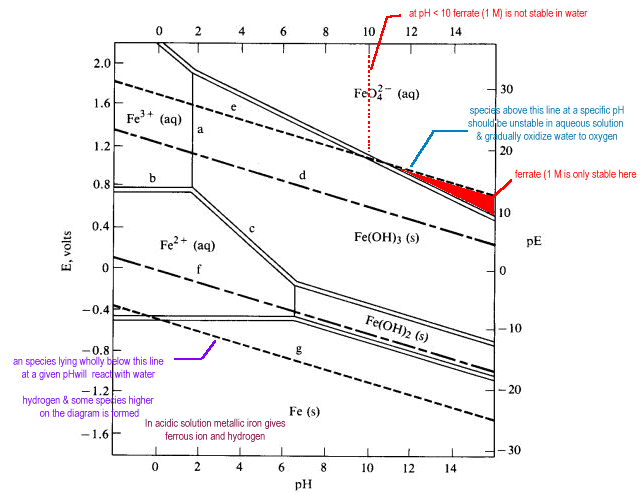
- Any point on the diagram will give the termodynamically most stable (theoretically the most abundant) form of the element for that E and pH.
- E=+0.8 V and pH = 14
predominant form is FeO42-.
- Strong oxidizing agents and oxidizing conditions are found ONLY at the top of the diagram.
The lower boundaries of strong oxidizing agents are high on the diagram. - Reducing agents and reducing conditions are found at the bottom of a diagram and nowhere else.
Strong reducing agents have boundaries that are low on the diagram. - A species that prevails from top to bottom at the pH in question has no oxidizing or reducing properties at all within that range.
| On the Pourbaix diagram for iron find: |
| Cations and anions will undergo redox reactions if the predominance area of their oxidation states do not coincide. |
Ferrate ion is expected to be a feebly basic anion which should precipitate with feebly acidic cations. Ferrate gives the expected precipitate with Ba2+ but not with Eu2+.
Eu2+ is a good reducing agent having no part of its predominance area above a potential of -0.429 V. There is no overlap of this region with that of ferrate ion. A redox reaction will occur between the two species to yield species that do have overlapping predominance areas -- Eu3+ and Fe3+
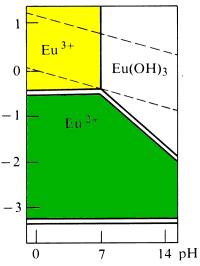
Pourbaix diagrams allow for more accurate predictions of the forms in which the different elements will exist in natural waters.
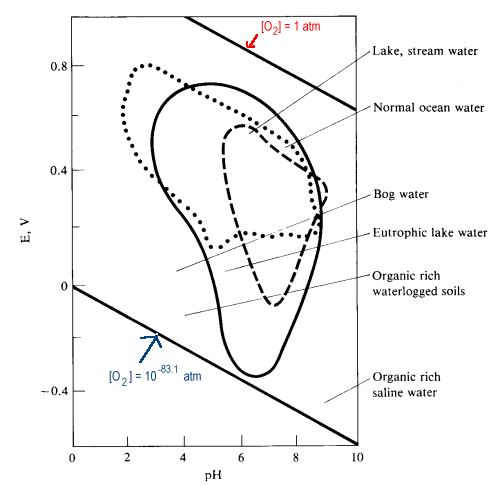
- For a clean lake, the surface waters are well aerated and the dissolved oxygen concentrations are high enough to make the potential reasonably close to the Eo for oxygen.
- Conditions may approach anaerobic (actively reducing)approaching the lower boundary of the reduction of water to hydrogen for
- a lake highly polluted with organic reducing agents
- the bottom layer of a thermally stratified lake
- for a swamp