 Lecture Notes on SN1 Reactions Lecture Notes on SN1 Reactions |
|
 | 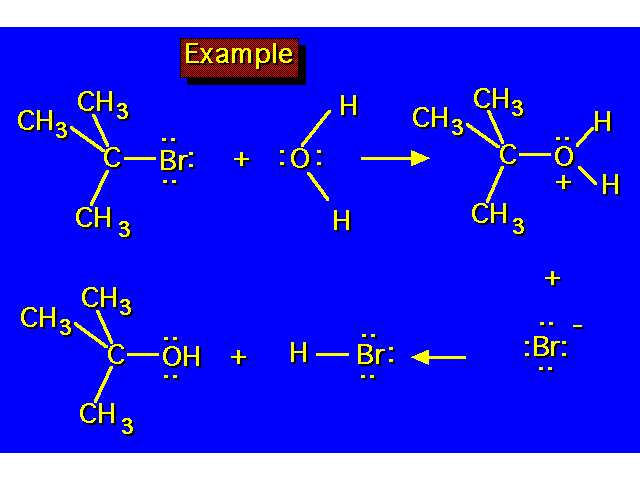 |
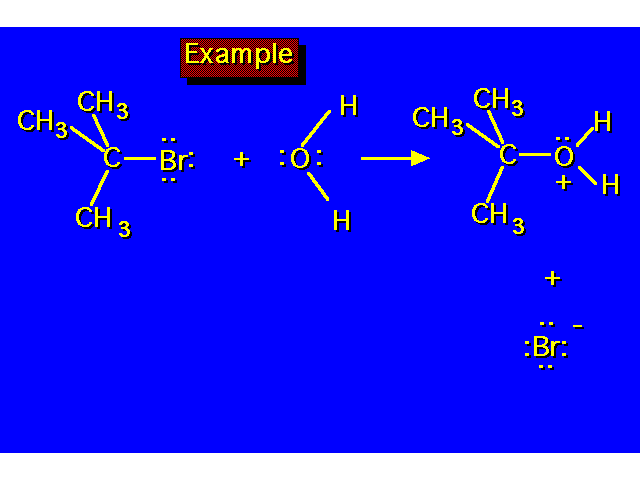 | |
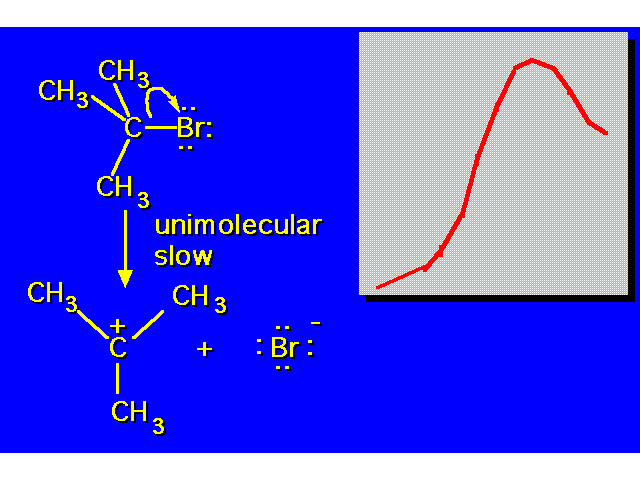 | |
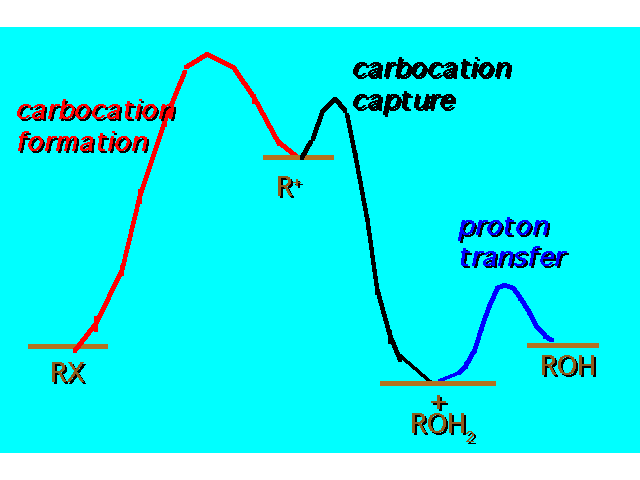 | |
 | |
 |  |
 | |
 | |
 |  |
 | 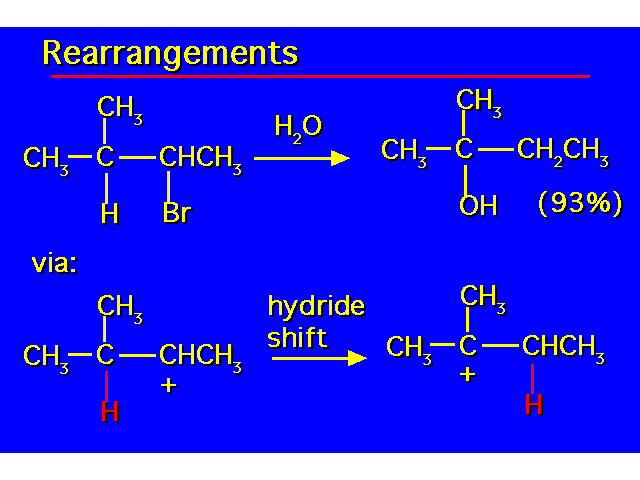 |
 | |
 |  |
| In SN1 reactions, polar solvents stabilize the developing charges via solvation, lowering the activation energy. In SN2 reactions, effects of solvent polarity isn't as significant. Polar protic solvents do not greatly enhance the reaction rate because they solvate the nucleophile. Polar aprotic solvents do cause some enhancement due to stabilization of the transition state. |  |
 | |

Direct suggestions, comments, and questions about this page to Arlene Courtney, courtna@wou.edu.
Last Modified January 09, 1998
Western Oregon University
Copyright © 1997 Western Oregon University