 Lecture Notes: Diels-Alder In Synthesis Lecture Notes: Diels-Alder In Synthesis |
|
| The clue for a Diels-Alder disconnection is a cyclohexene ring having an electron-withdrawing group on the side of the ring opposite to the double bond. | 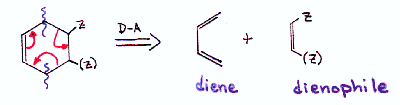 |
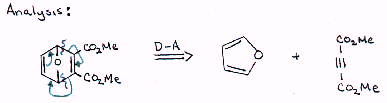 | |
 | |
| Ester groups are trans in the D-A adduct so they must be trans in the dienophile | 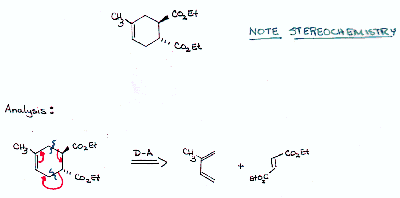 |
| Ha & Hbare cis in the product because they were cis in the quinone dienophile Hc is also cis to Ha & Hb because the D-A reaction is stereoselectively endo. | 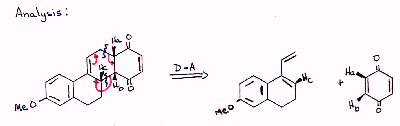 |
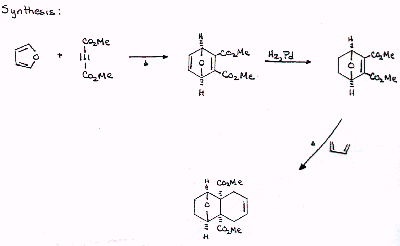
| Since the D-A reaction is such a good synthetic tool, it is wourth going to some trouble to get back to a recognizable D-A product in your analysis. Propose a synthesis of the target molecule that incorporates two Diels-Alder reactions. | 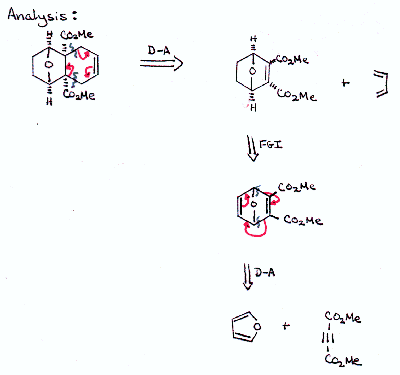 |
 | |

Direct suggestions, comments, and questions about this page to Arlene Courtney, courtna@wou.edu.
Last Modified February 1, 1998
Western Oregon University
Copyright © 1997 Western Oregon University