|
|
 |  |
 | |
 | |
| PROBLEM: Compound A has a molecular formula C7H12. Reaction of Compound A with cold aqueous KMnO4 gives Compound B (C7H14O2). When Compound A is treated with peroxybenzoic acid in chloroform, Compound C (C7H12O) is formed. Compound C reacts with dilute aqueous sulfuric acid to give Compound D (C7H14O2) which is isomeric with Compound B. Ozonolysis of Compound A gives 2,6-heptanedione. Assign structures to Compounds A, B, C, and D. 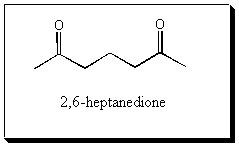 | |
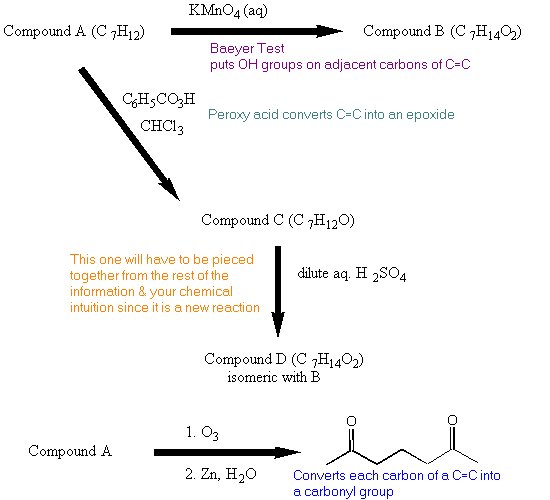 | |
| ANALYSIS: Look at the molecular formula for Compound A. Fully saturated formula would be C7H16. | two C=C two rings one C=C & one ring |
| Reaction of Compound A to form Compound B is the reaction in the Baeyer Test. | |
| Get the structure of Compound A from the ozonolysis data. | 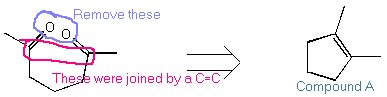 |
| The organic product in the Baeyer Test is a vicinal diol. This addition is a syn addition (This is new information for you). | 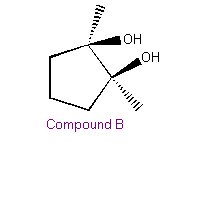 |
| Compound C is an epoxide. | 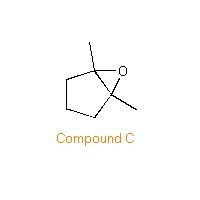 |
| Figuring out the structure of Compound D is a bit of a challenge since you have not yet studied any reactions of epoxides. However, you can use what you have learned so far this term to come up with a solution. Look at the reagents in the reaction:
Water can serve as a nucleophile. Compound D is an isomer of Compound B. | 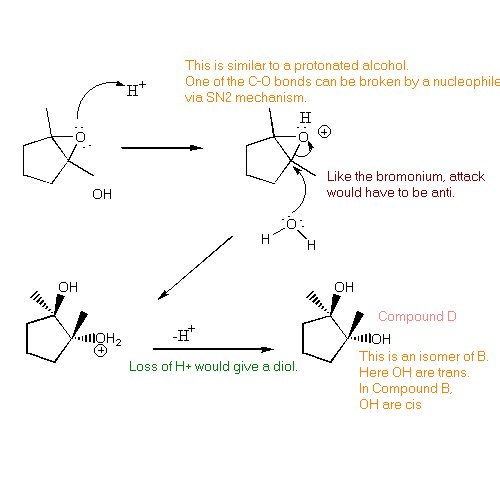 |
