|
|
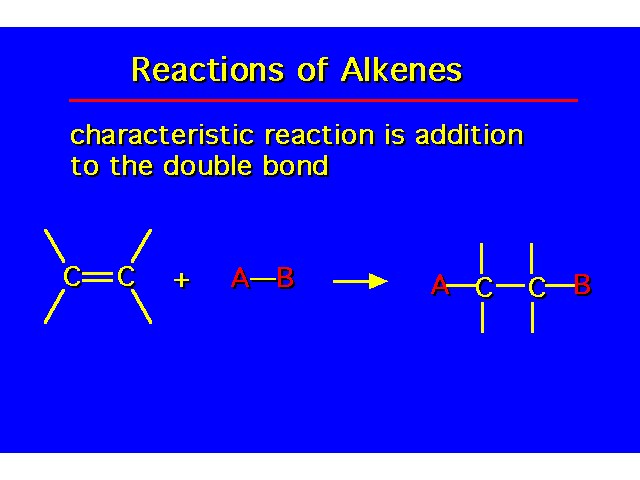 | |
| Hydrogenation of an alkene yields an alkane. | 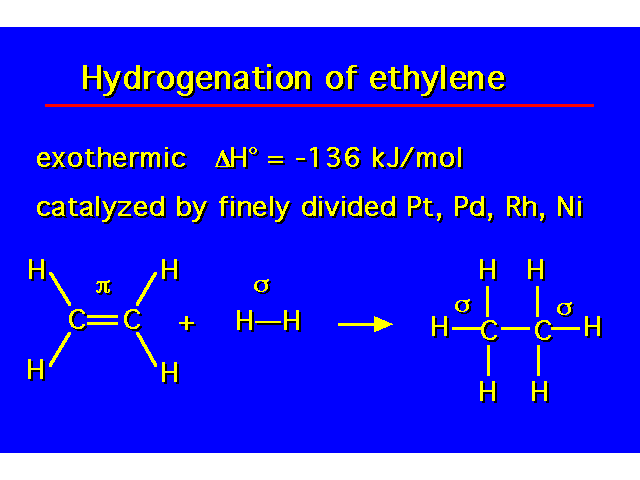 |
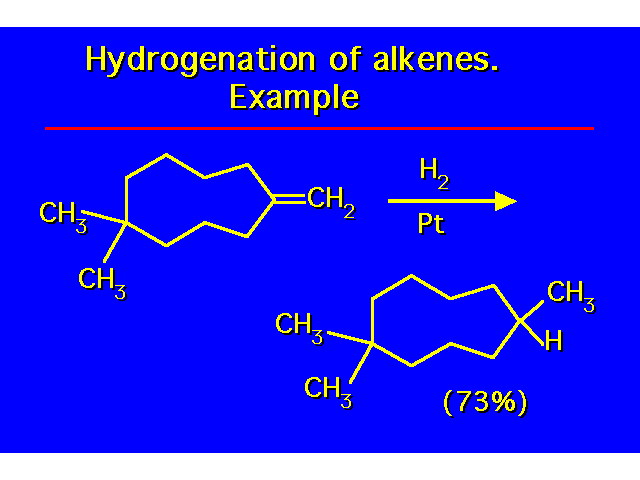 | |
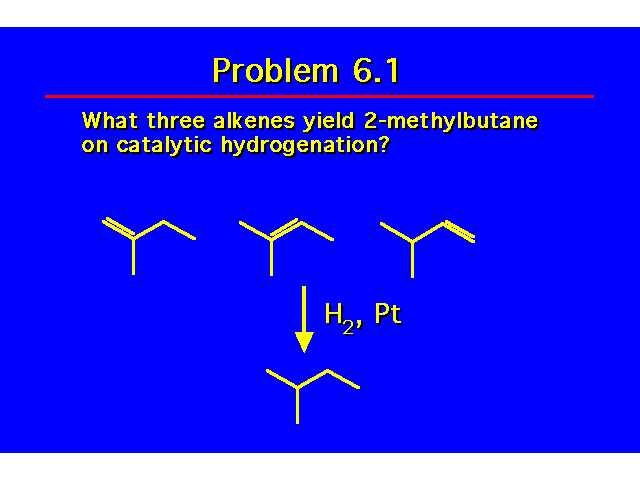 | |
| 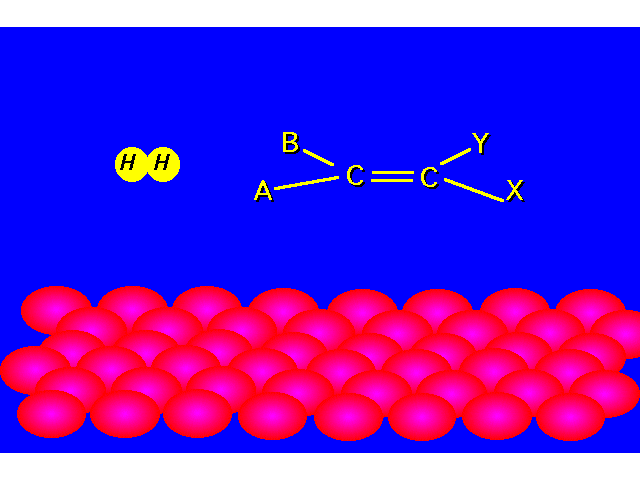 |
| STEP 1: Hydrogen molecules react with the metal atoms at the catalyst surface. The relatively strong H-H sigma bond is broken and replaced with two weak metal-H bonds. | 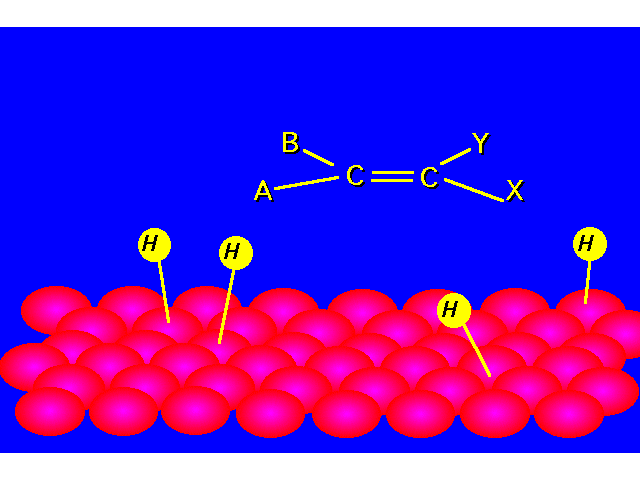 |
| STEP 2: The pi bond of the alkene interacts with the metal catalyst weakening the bond. A hydrogen atom is transferred from the catalyst surface to one of the carbons of the double bond. | 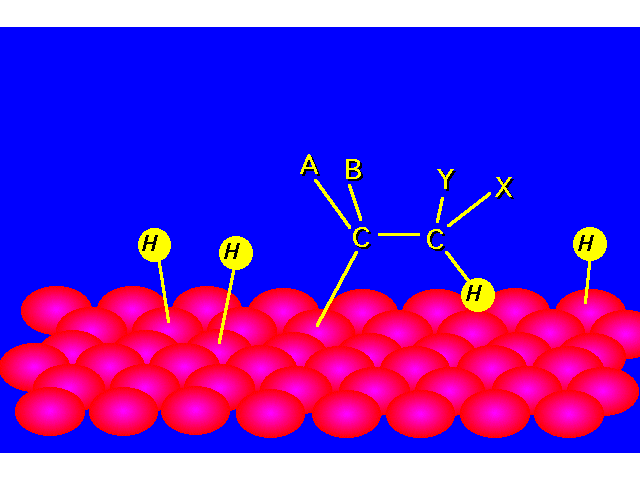 |
| STEP 3: The pi bond of the alkene interacts with the metal catalyst weakening the bond. A second hydrogen atom is transferred from the catalyst surface forming the alkane. | 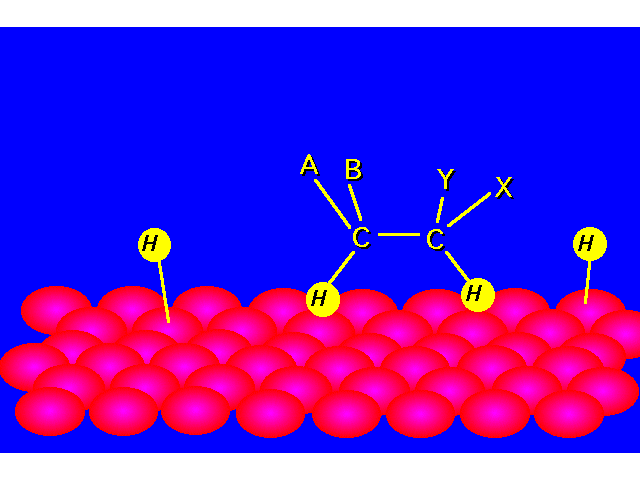 |
| The alkane is released from the catalyst's surface allowing the catalyst to accedpt additional hydrogen and alkene molecules. | 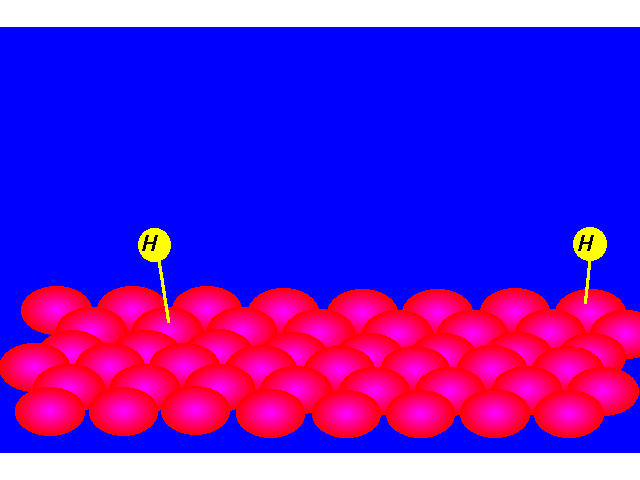 |
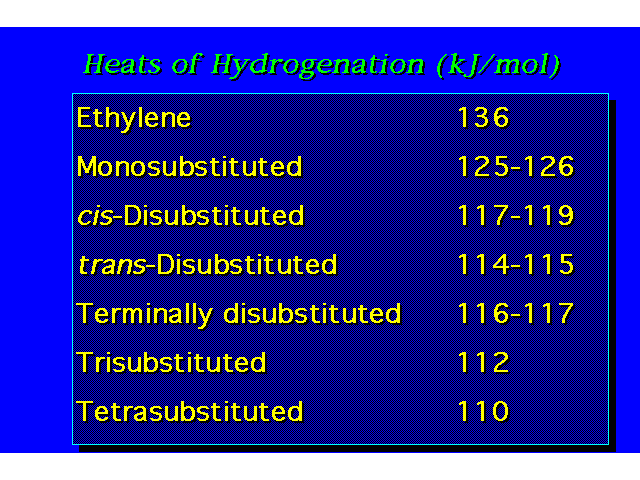
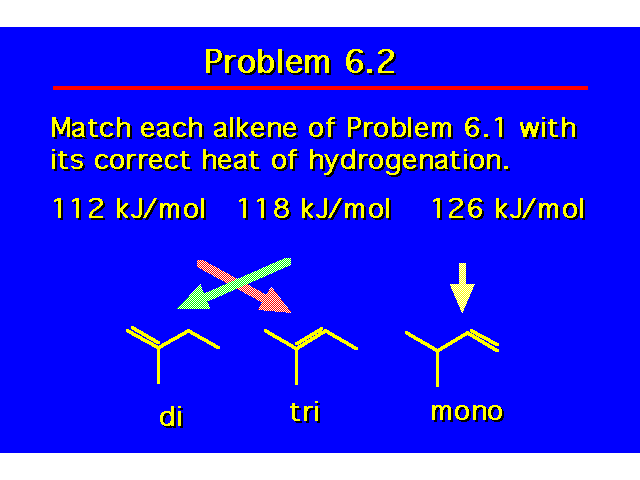
HEATS OF HYDROGENATIONHeats of hydrogenation correlate with structure just like heats of combustion do. The greater the heat of hydrogenation, the less stable the alkene. | 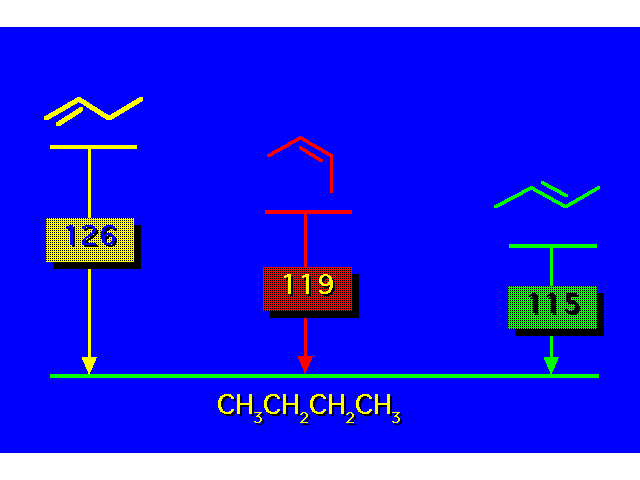 |
 | |
 | |
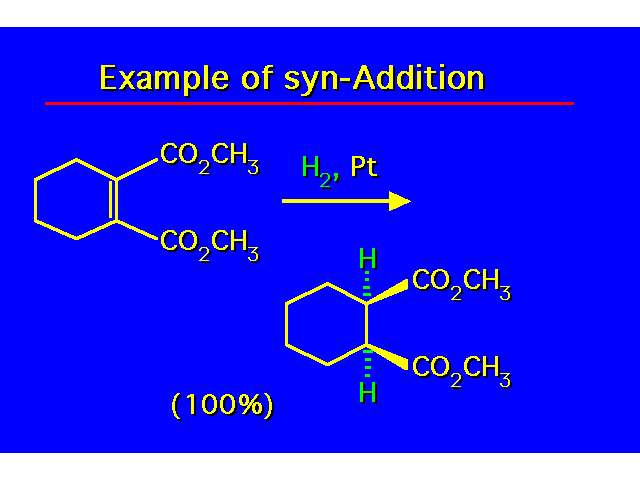
| In the addition both groups can add to the same face of the alkene (syn addition) or they can add to opposite faces (anti addition). Hydrogenation of alkenes is a syn addition. | 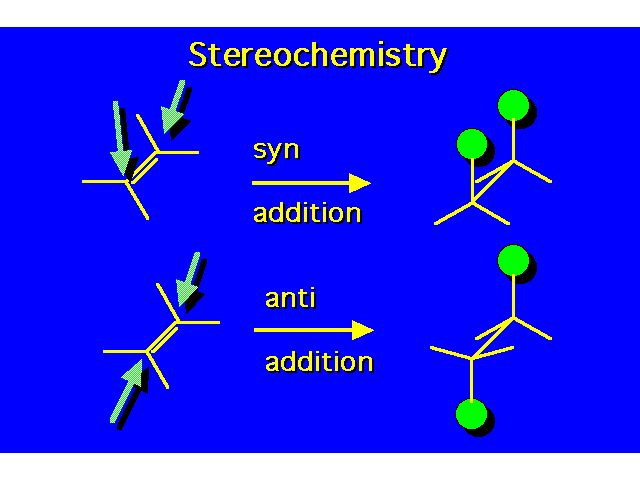 |
 | |
 | |
 | |
 | |
