|
|
 | |
| 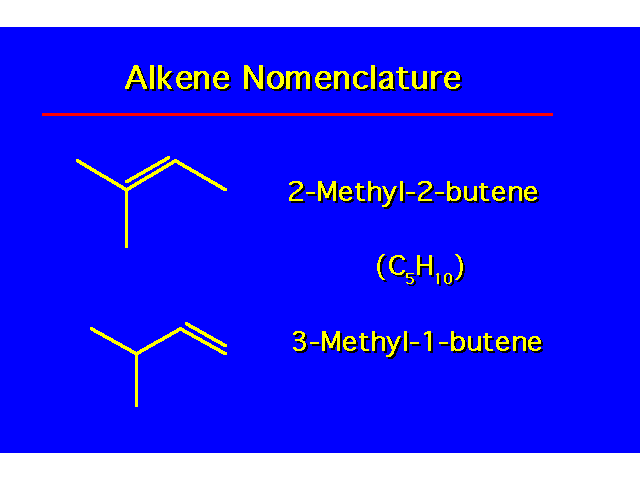 |
| The alkene functional group takes precedence over alkyl and halide functional groups. The alcohol functional group takes precedence over the alkene functional group. | 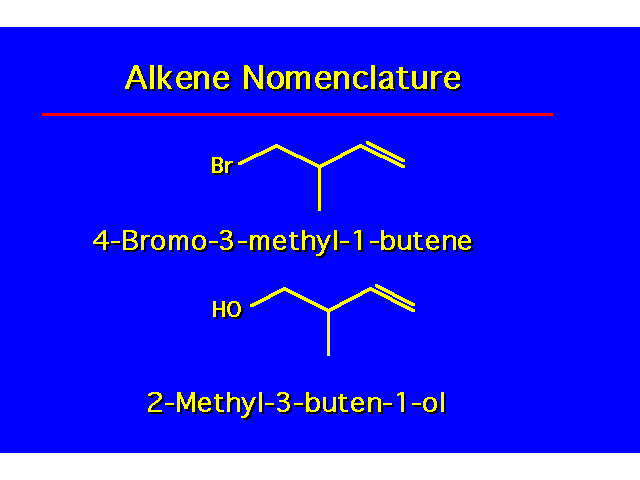 |
| To number the cycloalkene, one carbon of the C=C is given the number 1 and the other carbon of the C=C is given the number 2. Numbering is done to give the alkyl groups the lowest possible numbers. | 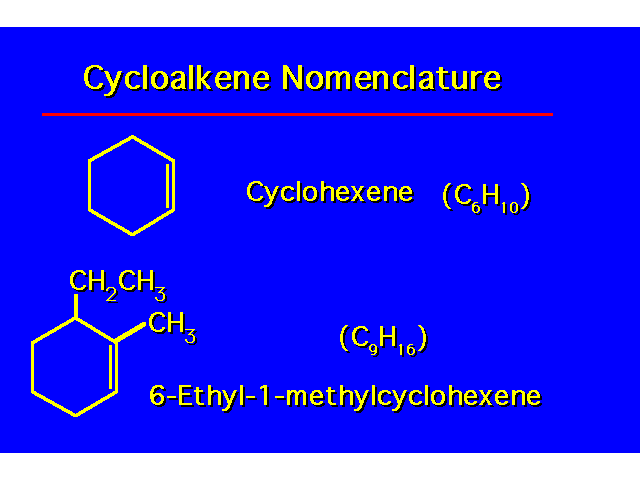 |
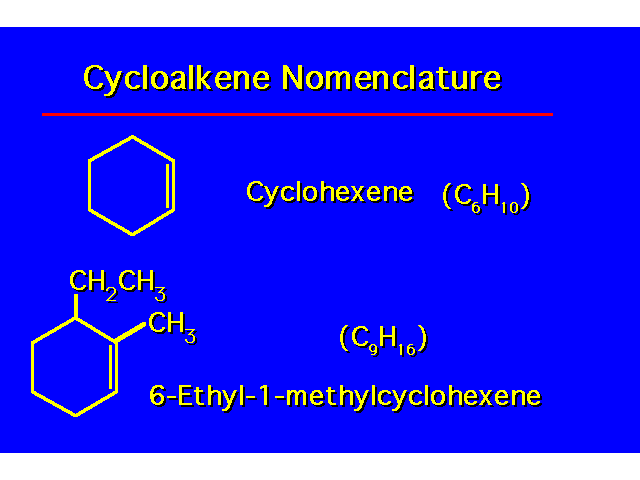 | |
 | |
 | |
 | |
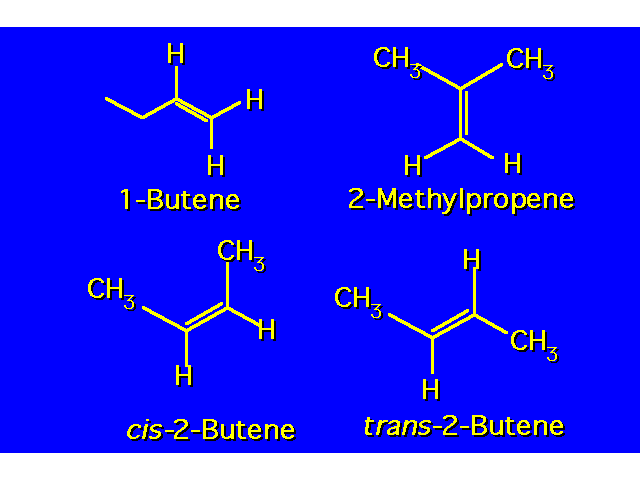 | |
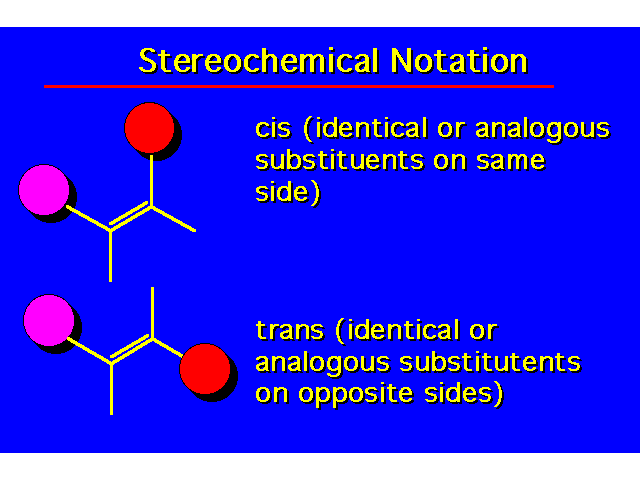 | |
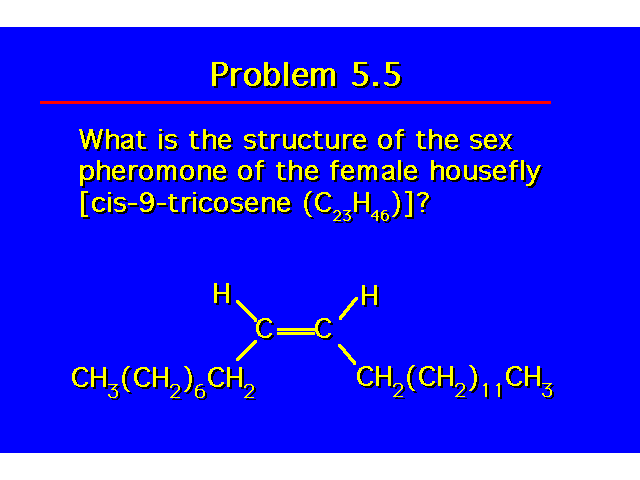 | |
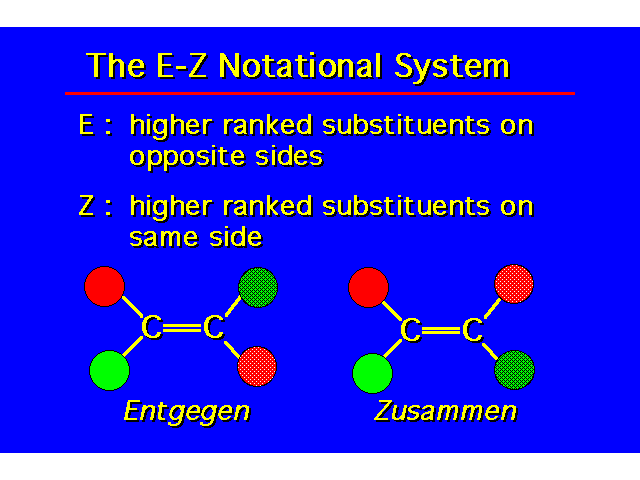
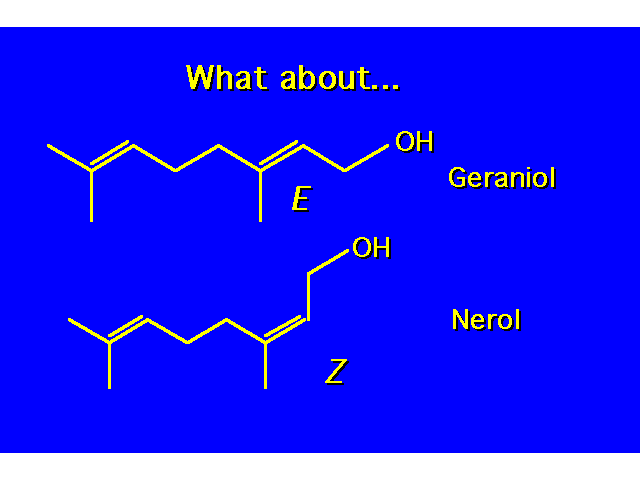
| The designation cis and trans are not unambiguous here. | 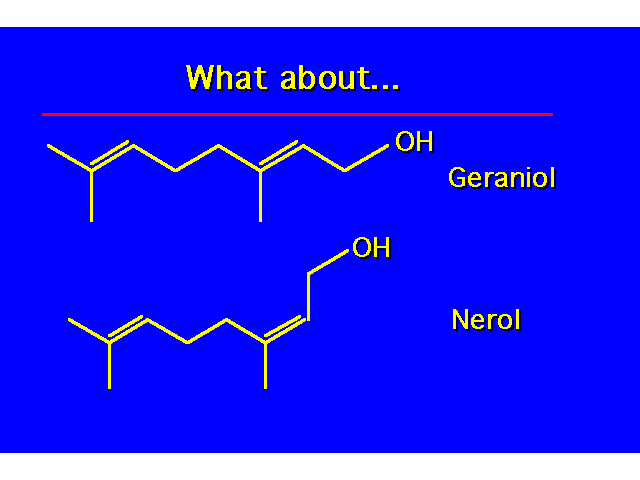 |
 | |
 | |
 | |
 | |
 | |
 | |
 | |
 | |
 | |
 | |
