|
|
|  |
| The enthalpy change for the reaction can be calculated from bond dissociation energies. | 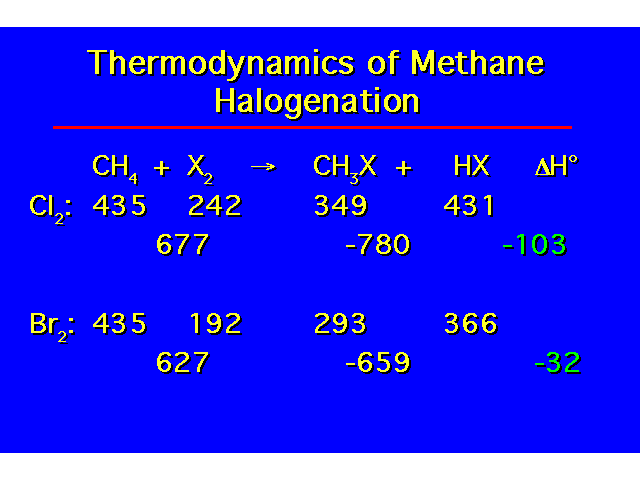 |
 | |
| During chlorination, polyhalogenation can occur. | 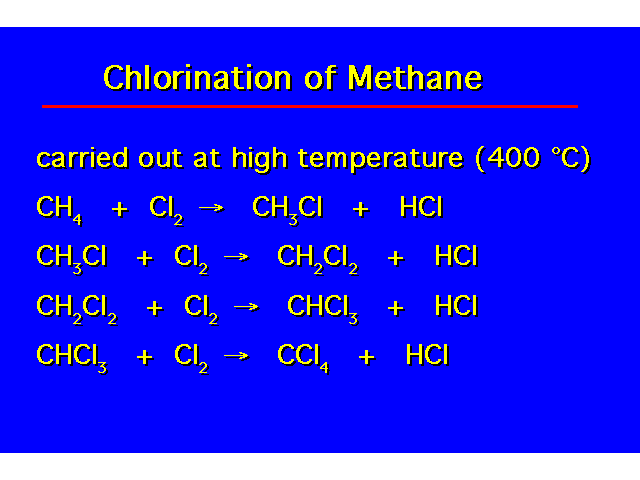 |
|  |
| Homolytic cleavage produces free radicals Heterolytic cleavage yields ions |  |
| A carbon free radical has characteristics similar to the carbocation. The geometry is planar with the unpaired electron in an atomic p-orbital. The species is electrophilic. |  |
 | |
 | |
| Alkyl groups stabilize free radicals for the same reasons that they stabilize carbocations. |  |
| Initiation Step: Initiates the reaction by generating a free radical reactant. This requires an energy input for homolytic bond dissociation to occur. A chlorine atom (free radical) must be generated to start the substitution process. This energy is generally supplied by either heating the reaction or by exposing the reaction to ultraviolet light. | 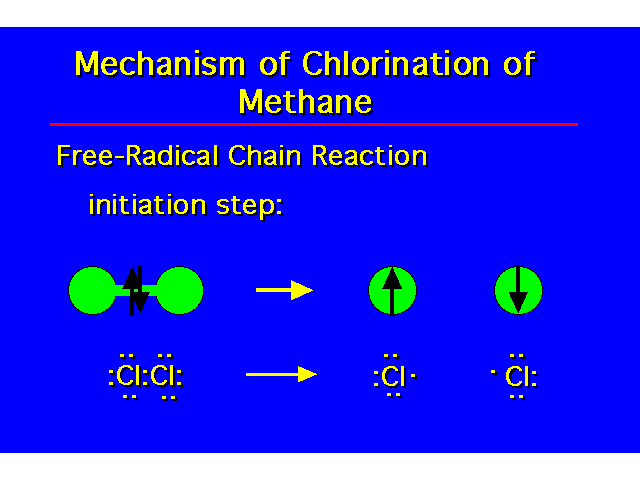 |
 | |
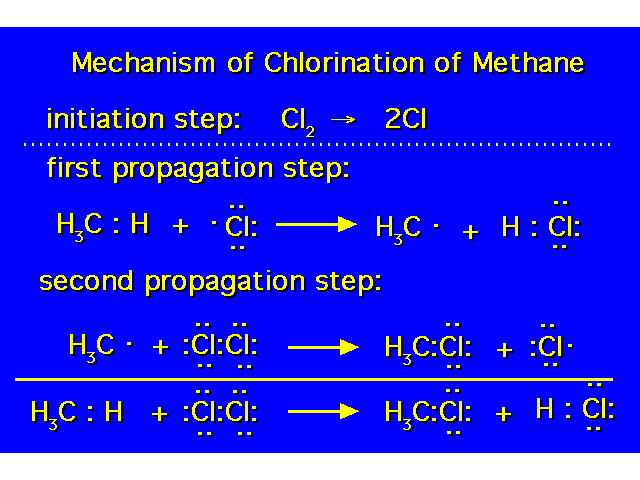 | |
| At the end of propagation step 2, a molecule of product has been formed along with a chlorine atom (free radical) that initiates another propagation step 1. | 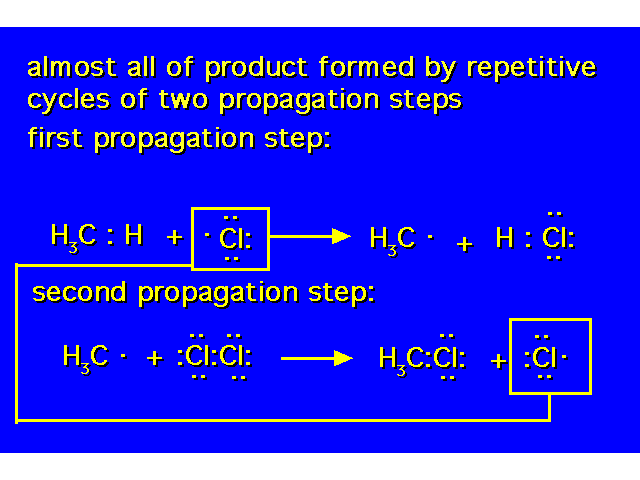 |
| Each time this step occurs, a chlorine free radical is removed that could have continued the chain while a methyl free radical is consumed that would have caused another chlorine free radical to be generated. |  |
| 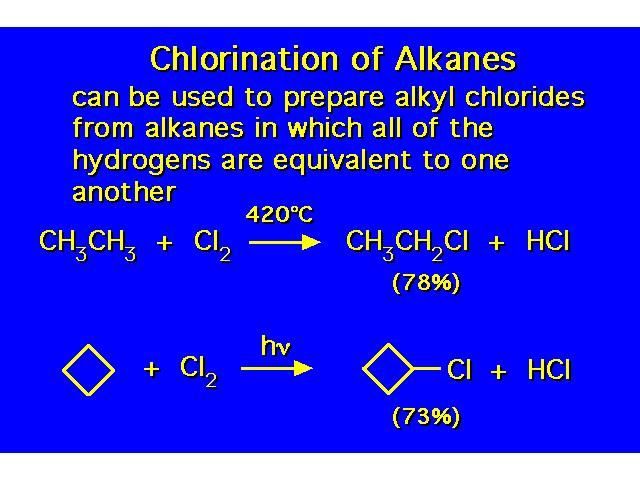 |
 | |
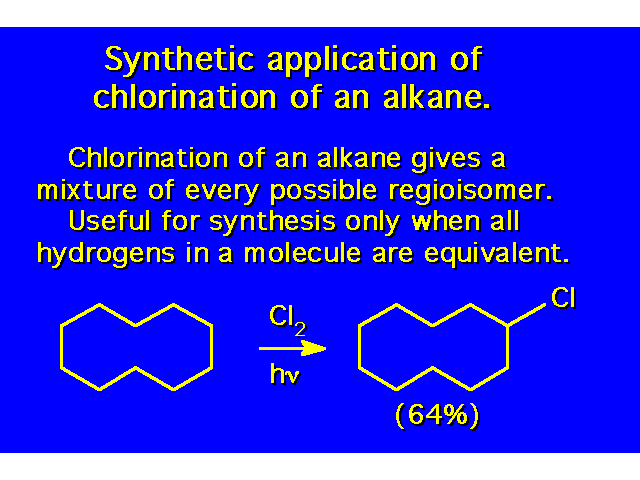
| Since there are 10 hydrogens in this molecule, you would expect that each hydrogen has a 10% chance of being the collision site. | 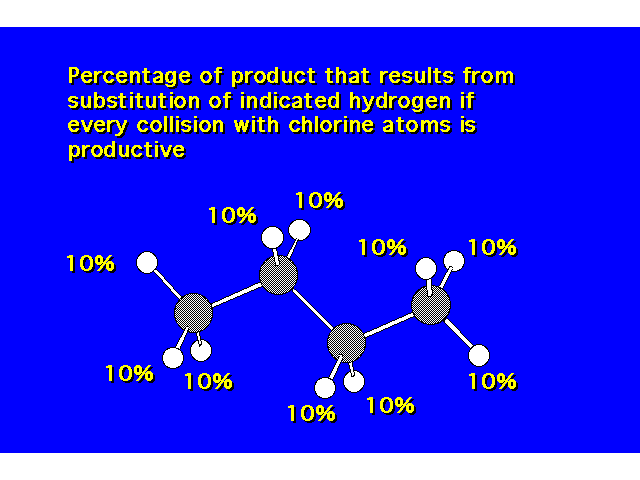 |
| Experimentally, the secondary hydrogens are found to be replaced more frequently than the primary hydrogens. | 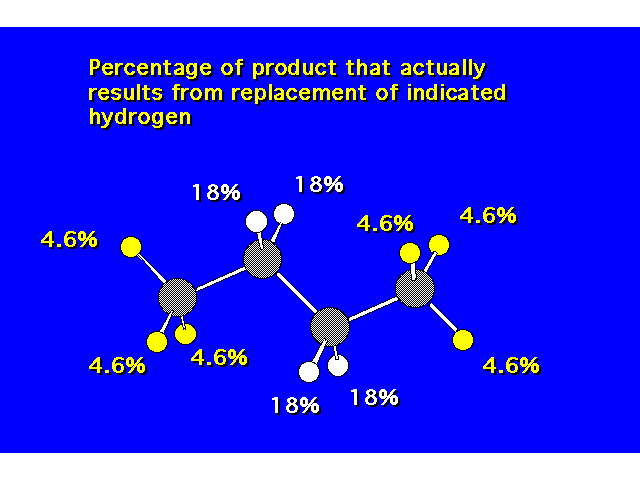 |
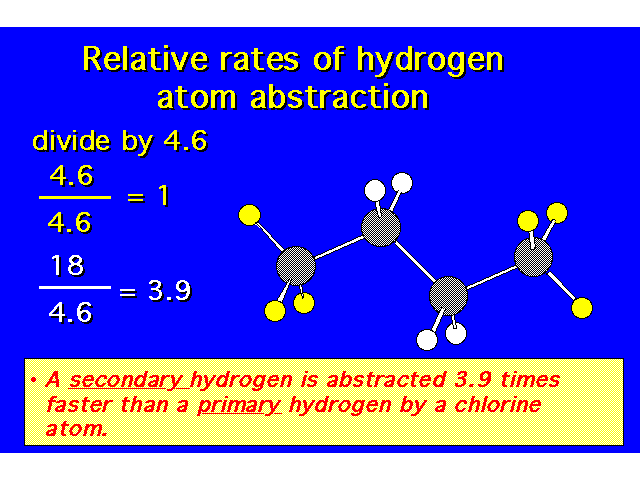 | |
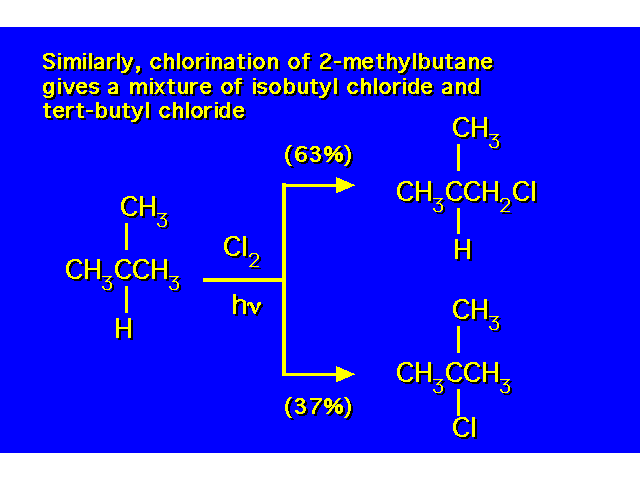 | |
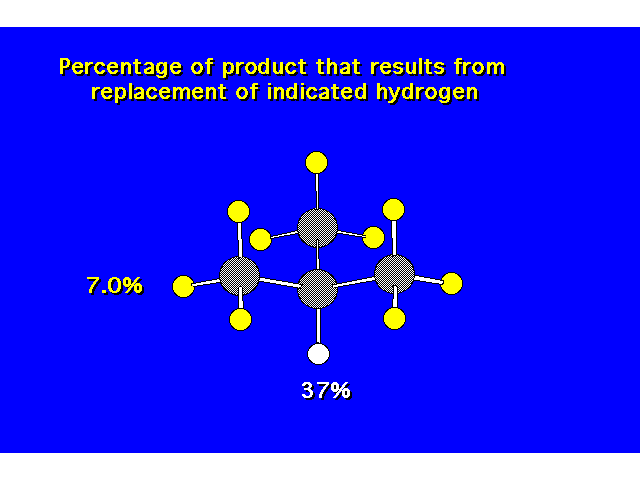 | |
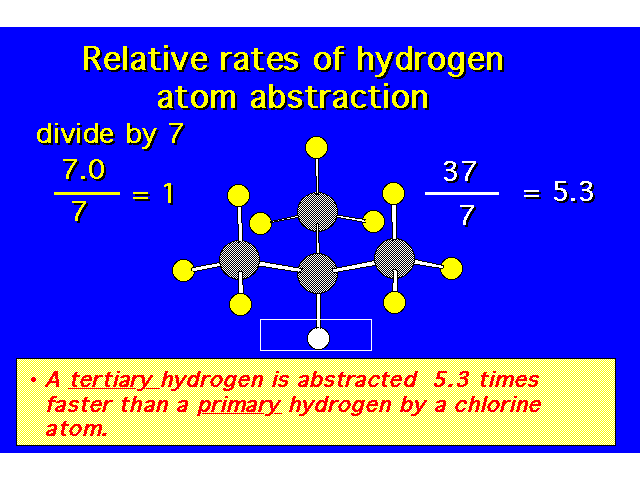 | |
 | |
 | |
 | |
 | |
