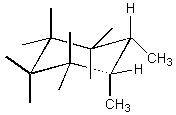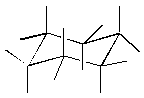Ch 334 Name_____________________________________
Midterm #2
November 21, 2001
1. (4 pts)
Consider the properties of the following compounds:
|
NAME
|
FORMULA
|
BOILING
PT (oC)
|
MOLECULAR
WEIGHT
|
|
Ethane
|
CH3CH3
|
-88.2
|
30
|
|
Fluoromethane
|
CH3F
|
-78.6
|
34
|
|
Methanol
|
CH3OH
|
+64.7
|
32
|
Why
does methanol boil so much higher than ethane or fluoromethane even though they
all have nearly equal molecular weights?
2. (2 pts)
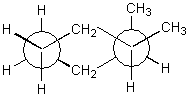
Consider the structure shown below.
Is the conformation given a cis or trans isomer?
3. (2 pts)
Complete the formula for the most stable conformation of
cis-1,3-dimethylcyclohexane. You only
need to draw in the groups that are at positions 1 & 3 as was done in
question #2.
4. (2 pts)
Consider the molecule shown at the right:
(a) Draw the other chair conformation of this
molecule.
(b) Circle the conformation above that is present
in greater concentration in the equilibrium
mixture.
5. (6 pts)
The following names are incorrect IUPAC names. Draw the structure implied by this name,
indicate why the given name is incorrect and write the correct name for each
structure.
(a) 2-methylcyclohexene
(b) 1,2,2-trichloro-4-pentene
6. (4 pts)
In the equilibrium below, the strongest acid is __________________.
H2SO4 + (CH3)2CHOH D HSO4- + (CH3)2CHOH2+
7. (6
pts) Give IUPAC names for the following
compounds:
(a)
_________________________________________________
(b)
_________________________________________________
8. (6 pts)
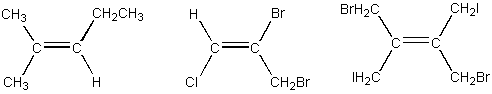
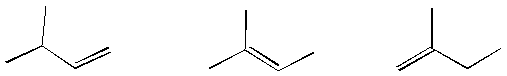
Circle any of the alkene structures shown below which have geometric
isomers.
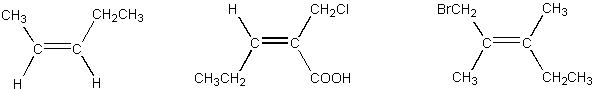
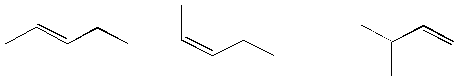
9. (6 pts)
Assign E/Z configurations to each of the following alkenes.
_______ _______ _______
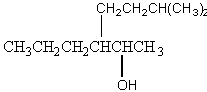
10. (6 pts)
Give the alkenes that can form from acid-catalyzed dehydration of the
following alcohols. If more than one
product can form, identify the products as major or minor.
(a)
(b)
11. (4 pts)
Although you have not seen reactions involving the following species,
your background knowledge concerning nucleophiles and electrophiles is enough
to answer this question. Indicate whether the following species would be
expected to react as nucleophiles or electrophiles. Justify your choice.
(a) :Cl+ __________________
Reason:
(b) (CH3)3C:- __________________ Reason:
12. (8 pts)
Identify the Species I and II as electrophiles (E) or nucleophiles (Nu)
in the reactions:
Species
I Species II
(a) (CH3)2O: + B(CH3)3 ---------> (CH3)2O-B(CH3)3
_______ _______
(b) :CN:- + CH3Br ----------> H3C:CN
+ :Br:-
_______ ________
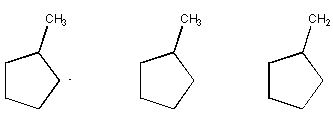
13. (16 pts)
Circle the compound in each group that fits the description provided.
(a) Has the lowest Heat of Hydrogenation
(b) Has the highest Heat of Combustion
(c)
Is lowest in energy.
(d) Is most stable.
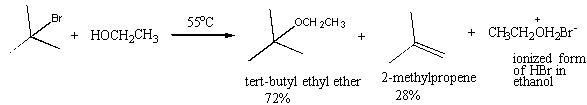
14. The reaction of tert-butyl bromide with
ethanol shown below is termed a solvolysis reaction and gives the products
formed.
Experiment
shows that when the concentration of t-butyl bromide is doubled without changing the amount of ethanol, the
rate of reaction doubles. When the
concentration of tert-butyl
bromide is held constant and the concentration of ethanol doubled, no change in
the reaction rate is observed.
(a) (6 pts) Draw a mechanism showing how
2-methylpropene forms that is consistent with the kinetic data presented
above. DO NOT COMBINE STEPS IN YOUR
MECHANISM. You must use arrows to show
all electron movement.
(b)
(4 pts) Draw a potential energy diagram
depicting the intermediates and transition states involved in the
reaction.
(c)
(2 pts) What is the rate determining step of the reaction?
15.
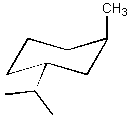
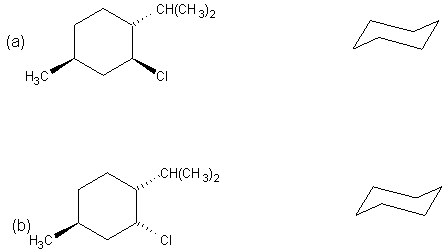
(6 pts) Draw the
most stable chair conformation for the two molecules shown below. One of these
stereoisomers undergoes elimination on treatment with sodium ethoxide in
ethanol much more readily than the other.
Circle the one which reacts faster.
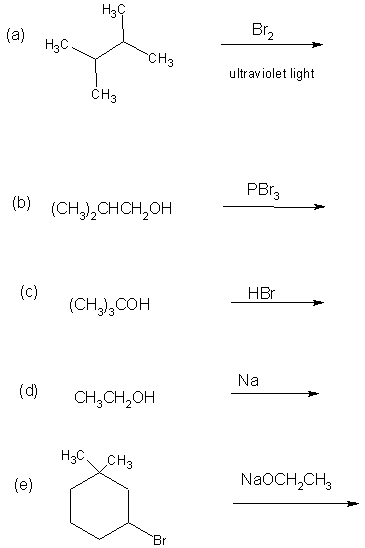
16. (10 pts)
Complete the following reactions by showing the MAJOR organic product.
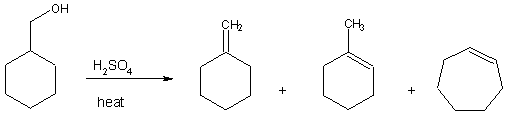
Bonus
Question (10 pts):
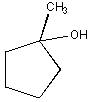
Dehydration
of cyclohexylmethanol in the laboratory was found to yield three different
alkene products. One of these products was cycloheptene. Draw a mechanism that clearly shows how this
product forms.