|
Gas Hydrates
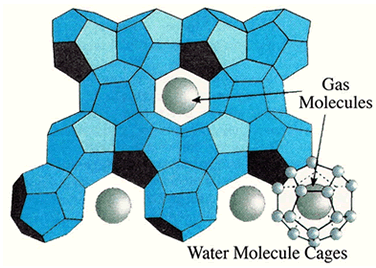
Gas hydrates are crystalline solids in which gas molecules are encased in cages of water molecules. The solid is similar to ice except that it burns! The gas molecules, called a guest, actually stabilize the solid structure in these hydrates. There are a number of gases that have molecular sizes appropriate to being guests in hydrate structures including carbon dioxide, hydrogen sulfide, and some low-carbon-number hydrocarbons.
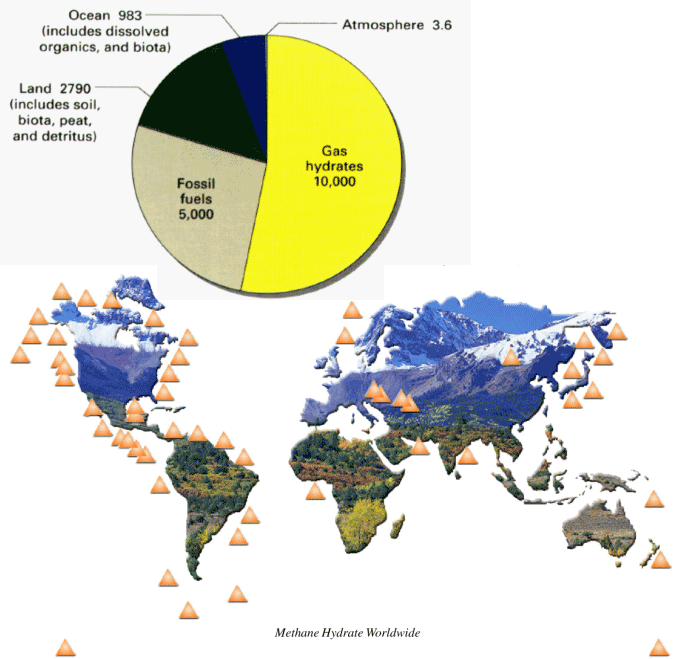
Methane hydrates are found primarily in two types of cold environments, deep in the ocean in deep water continental margins and beneath the permafrost in arctic regions. The methane itself is the product of microbial action on decaying organic matter that becomes trapped in an ice matrix. These hydrates (or clathrates as they are sometimes called) are found mixed with sediment. Source: United Nations Environment Programme
diagram source: Energy Technology Laboratory This video addresses the question: "Why are methane gas hydrates important for society?". This video discusses some possible environmental issues concerning gas hydrates.
Watch this video to learn more about the research being done in Inuvik of the Canadian Northwest Territories on hydrates (click here to see where Inuvik is located).
Experts believe that Earth's gas hydrate reserves far exceed the remaining quantities of other fossil fuels. The diagram below shows those regions where gas hydrates are believed to exist.
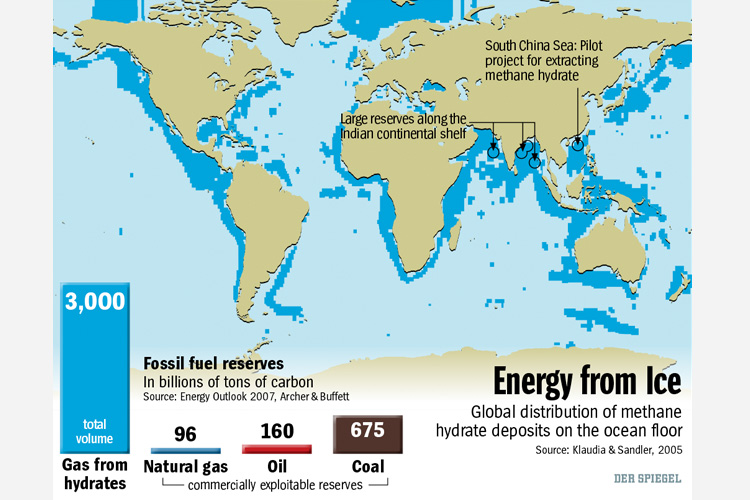
source: Los Alamos National Laboratory India and China are two countries that have to import large amounts of petroleum products. Read this Der Spiegel Online International article discussing how China and India are trying to exploit methane hydrates.
Since Japan has to import nearly all of the fossil fuels that it uses and in the aftermath of the Fukushima Daiichi nuclear disaster has taken most of it's nuclear reactors off line, it is looking for new energy sources. On March 12, 2013, the Japanese Ministry of Economy, Trade and Industry announced the first successful extraction of methane from a gas hydrate deposit 1,000 feet below the seabed in the Eastern Nankai Trough off the southeastern coast of Japan. Time will tell if this can be developed into a commercially viable natural gas source. More locally, recent discoveries indicate that subduction of the Juan DeFuca plate has scraped away sediments exposing significant accumulations of hydrates off the Washington and Oregon coasts. Alaska is also believed to have vast supplies of methane hydrates that would far exceed the amount of oil produced from the north slope and other regions.
Learn more about "frozen fuel" here.
Page last modified 14 November 2019 |


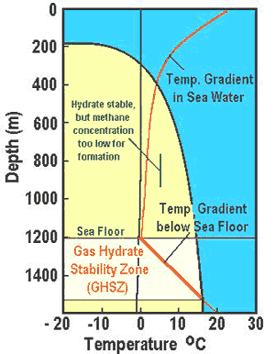 These hydrates need low temperatures and high pressures to form. In the ocean, the temperature decreases with water depth to almost 0°C at the bottom. Temperatures begin to increase again as you decent beneath the ocean floor. The gas hydrate stability zone (GHSZ), the conditions of ocean depth under which hydrates are stable, can be seen in the diagram to the right. For this example the sea floor is at a depth of 1200 meters. Although the stability of hydrates starts about 400 meters, hydrates only accumulate in the sediments or as mounds on the seafloor directly over point sources of methane release. Watch this video about hydrates on the ocean floor:
These hydrates need low temperatures and high pressures to form. In the ocean, the temperature decreases with water depth to almost 0°C at the bottom. Temperatures begin to increase again as you decent beneath the ocean floor. The gas hydrate stability zone (GHSZ), the conditions of ocean depth under which hydrates are stable, can be seen in the diagram to the right. For this example the sea floor is at a depth of 1200 meters. Although the stability of hydrates starts about 400 meters, hydrates only accumulate in the sediments or as mounds on the seafloor directly over point sources of methane release. Watch this video about hydrates on the ocean floor: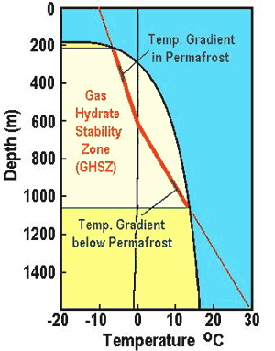 The figure to the left shows the type of conditions needed for hydrates to form in arctic permafrost regions (in this example the depth of of permafrost is 600 meters). The GHSZ extends from approximately 200 meters to about 1,000 meters.
The figure to the left shows the type of conditions needed for hydrates to form in arctic permafrost regions (in this example the depth of of permafrost is 600 meters). The GHSZ extends from approximately 200 meters to about 1,000 meters.